CAR T Cell Therapy
Table of Contents
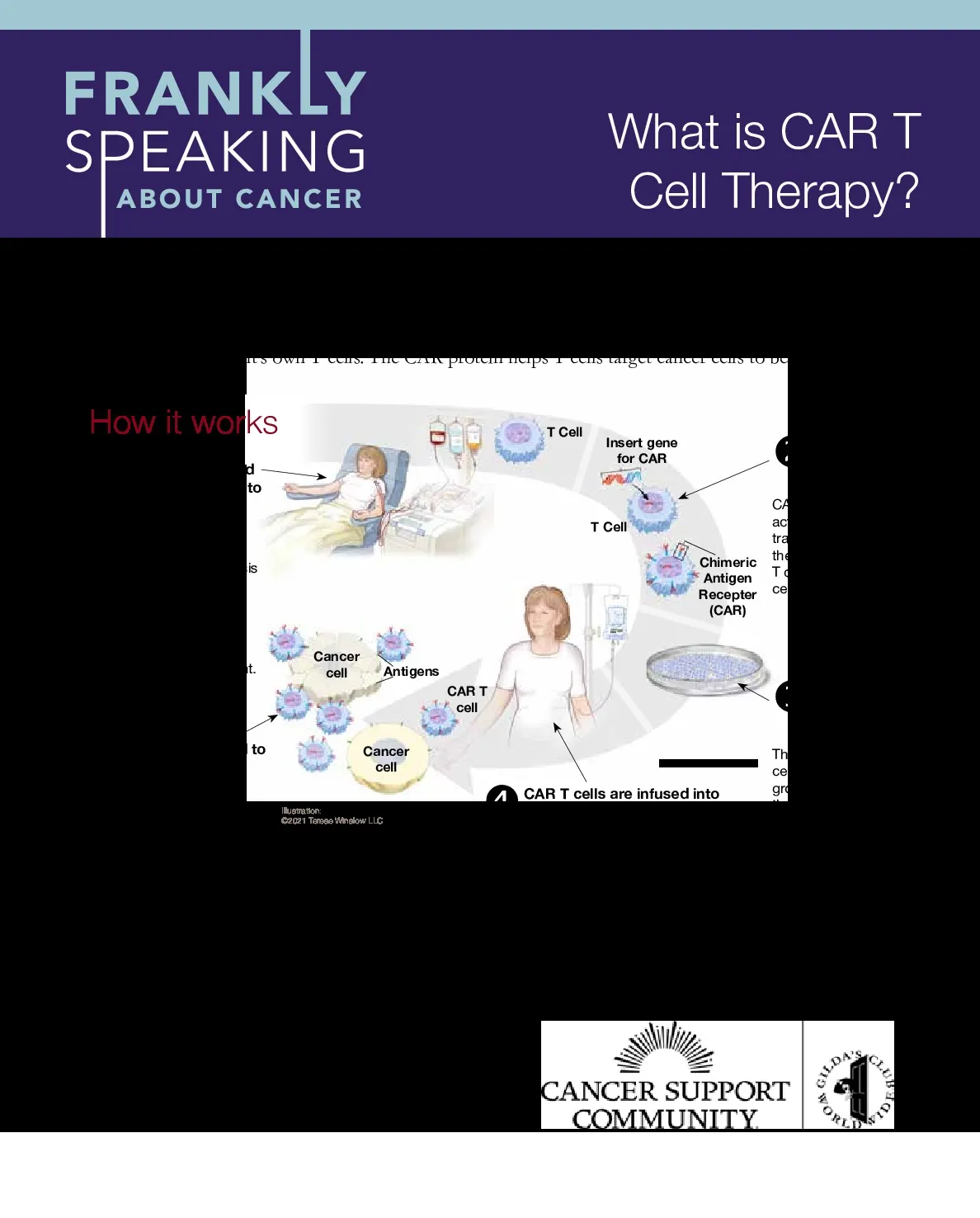
CAR T cell therapy, or chimeric antigen receptor T cell therapy, is one way to use the body’s natural defenses to fight cancer. It is called CAR T cell therapy because a lab-made protein, called chimeric antigen receptor (CAR), is added to the patient’s own T cells. T cells are a type of white blood cell found in the body. The CAR protein helps T cells recognize the cancer cells to be killed. This protein also helps train your immune system, which is your body’s natural defense against disease, to attack cancer cells. T cells are your immune system’s “fighters.” Their job is to seek out and attack viruses, bacteria, and unhealthy cells, like cancer.
What is Cell Therapy?
Ruby, your virtual community navigator, will help explain what you need to know about cell therapy and how it is used in cancer treatment.
CAR T cell therapy, or CAR T, is a type of cancer treatment called immunotherapy. Immunotherapies work with the body’s immune system to find, attack, and kill cancer cells. These drugs can work in different ways. Some strengthen the immune system, so it can fight cancer better. Other immunotherapy drugs help the immune system better recognize cancer cells.
The U.S. Food and Drug Administration (FDA) approved the first CAR T therapy in 2017.
CAR T is an approved treatment option for a variety of blood cancers, like multiple myeloma and certain types of leukemia and lymphoma.
CAR T Webinar Part 1: Sustaining Compassionate Care
Guest speakers Brittany Fols, LCSW, OSW-C and Lauren Kriegel, LCSW, OSW-C, provide practical tips to help cope with the social and emotional challenges of going through CAR T cell therapy, including the importance of self-care. Self-care strategies…
Who Can Get CAR T?
To date, the U.S. Food and Drug Administration (FDA) has approved 6 CAR T cell therapy treatments. These treatments are typically for patients where the cancer is refractory or relapsed after having 1 or more prior treatments. Refractory means the cancer did not respond to the previous treatment. Relapsed means the cancer returned even after prior treatments. New CAR T cell therapy treatments and new ways to use currently approved treatments are being studied in clinical trials.

Step-by-Step Support
Our guide for CAR T patients and caregivers provides support through each stage of the CAR T process, from referral to post-treatment care.
Guía para cuidadores y pacientes
Esta guía proporciona información paso a paso desde el inicio del tratamiento hasta el proceso de seguimiento.
CAR T Cell Therapy Quick Guide
Welcome to this short guide on CAR T Cell Therapy. It will explain what CAR T cell therapy is and which types of patients it might help. CAR T cell therapy is one type of immunotherapy. Immunotherapies use your body’s immune system to identify,…
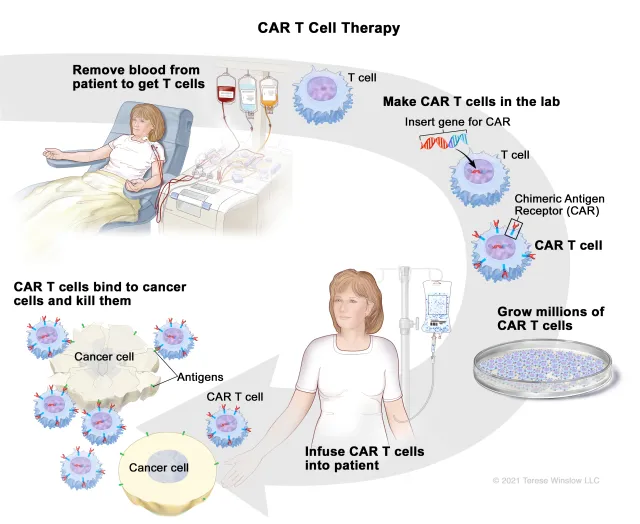
How Does CAR T Work?
CAR T cell therapy removes some of a patient’s T cells and alters them to make them better able to fight cancer. The altered T cells are then returned to the patient’s body to go to work. CAR T cells harness the power of a patient’s immune system to fight against the cancer.
Here’s how the CAR T cell therapy process works:
- Blood is removed from the patient to get the T cells – T cells are collected from your bloodstream in a procedure called apheresis. The T cells may be frozen and kept in a special temperature-controlled storage unit until the CAR protein is added.
- CAR T cells are made in the lab – CAR proteins, which act like cancer-cell tracking devices, are then added to your T cells. Now your T cells are CAR T cells.
- Millions of CAR T cells are grown – The new CAR T cells then multiply and grow in the lab until there is enough for your treatment.
- CAR T cells are infused into the patient – Once the CAR T cells are ready, they are infused back into your bloodstream. You may get a brief course of chemotherapy (referred to as conditioning chemotherapy) before getting the CAR T infusion. This is done to destroy regular T cells and give the CAR T cells more room to operate. This also helps to increase the treatment’s effectiveness.
- CAR T cells bind to cancer cells and kill them – Once the CAR T cells are in your bloodstream, they attack the cancer cells.
CAR T is designed to be a one-time treatment. CAR T cells can remain in the body and continue to be active for a long period of time. After you get the new CAR T cells, you will need a caregiver to be with you 24 hours a day, 7 days a week to support and watch for side effects. The length of time you will need to have a caregiver with you to monitor for side effects can vary.
CAR T Patient Journey
Laura's CAR T Story
Laura, a survivor of multiple myeloma, talks about her experience with CAR T cell therapy.
CAR T: Caregiver Stories
Caregivers talk about their perspective and experience during CAR T cell therapy.
Lisa's CAR T Story
Lisa, a survivor of diffuse large B-cell lymphoma (DLBCL), talks about her experience with CAR T cell therapy.
Maxine and Dave's CAR T Story
Maxine, a survivor of non-Hodgkin lymphoma, and Dave, her caregiver, talks about their experience with CAR T cell therapy.
Scott's CAR T Caregiver Story
Scott, a caregiver, talks about his and Robin's, a survivor of diffuse large B-cell lymphoma, CAR T experience.
Making the Decision to Have CAR T
Patients and caregivers talk about their decision to have CAR T.
Kristin and Benny's CAR T Story
Kristin, a survivor of acute lymphoblastic leukemia (ALL), talks about her CAR T experience along side her caregiver Benny.
Shahzad and Nicole's CAR T Story
Shahzad, a survivor of diffuse large B-cell lymphoma (DLBCL), talks about his CAR T experience alongside his caregiver Nicole.
Parenting With Cancer: CAR T
Patients and caregivers talk about their experience parenting with cancer.
Jose's CAR T Caregiver Story
Jose talks about his experience as a caregiver during CAR T.
Kristin and Benny's CAR T Story
Kristin, a survivor of acute lymphoblastic leukemia (ALL), talks about her CAR T experience along side her caregiver Benny.
Where Is CAR T Given?
At first, CAR T treatments were only available at a few cancer centers. Now more than 150 cancer centers in the United States offer CAR T cell therapy. Your oncologist may work at a cancer center that has a CAR T program. If not, they may refer you to a program at another cancer center. This center may be near you or in another city or state. To get CAR T, you may need to be away from home for several months at a time.
- Referral to CAR T: Your primary oncologist refers you to a CAR T program to discuss an FDA-approved CAR T therapy or a clinical trial. If you seek a second opinion at a cancer center, you may learn of CAR T that way.
- First Appointment: Your first appointment allows you to learn more about the cancer center’s CAR T program. This appointment will also help the cancer center decide if CAR T is right for you. The treatment team will do some screening tests to make sure you are eligible and healthy enough to go through the treatment.
- Screening Tests: During your first appointment with the CAR T team, or soon after, you may have screening tests to see if this is a good treatment option for you. Additional screening tests may be broken into multiple appointments during your first visit. Timing of your first appointment and screening tests may vary based on the center’s availability and the healthcare provider’s schedule.
- Treatments to Qualify for CAR T: You may need treatment to become healthy enough to participate in CAR T therapy. These kinds of therapies may cause month-long waits before you start the CAR T therapy process. Even if you are healthy enough at this point in the process to start CAR T, you may need bridging treatment to stay healthy long enough to get to the “infusion” step of CAR T therapy.
- Apheresis: During apheresis you will be hooked up to a machine where your T cells will be separated from your other blood cells. After apheresis, your T cells will be sent to the lab where the CAR protein will be added. The apheresis procedure will take a few hours.
- Bridging Therapy: Bridging therapy is cancer treatment given at any step before CAR T infusion to keep you healthy enough to get to infusion. You may need to get bridging therapy multiple times throughout your journey, or you may not need bridging therapy at all. Each round of bridging therapy can last 1 or more months, but in some cases, it may only span 1-2 weeks.
- Conditioning Chemo: Before CAR T infusion, you will have conditioning chemotherapy (also known as lymphodepleting chemotherapy) to make room for your new CAR T cells. Conditioning chemotherapy is typically given over 2-3 days with 2-3 rest days. However, the timeframe may vary if you are in a clinical trial.
- CAR T Cell Infusion: At infusion, the new CAR T cells will be infused into your bloodstream. The CAR T cells will begin to attack the cancer cells in your body. Depending on your CAR T treatment, you may need to go to the hospital to receive the infusion. If it can be done in an outpatient clinic, the infusion appointment may take 20-30 minutes based on your healthcare team and facility.
- First 4-8 Weeks After Infusion: CAR T infusion can cause side effects. You may have to stay in the hospital for up to 1 week following the infusion. You will also need to be monitored closely by your caregiver and stay near the treatment center for at least 4 weeks after infusion.
- Weeks 9-52 After Infusion: You will need to follow up with the CAR T treatment team, your primary oncologist, or other specialists that understand your unique needs after the treatment is complete. Most often, you will have monthly appointments for the first year after infusion. Then, you may have annual appointments to monitor for any new cancers.

Contact Our Helpline
If you have questions or need additional support at any point on your CAR T journey, reach out to the CSC’s CAR T Cancer Support Helpline at 844-792-6517.
Side Effects of CAR T
The CAR T infusion can cause side effects. For at least 4 weeks after the infusion, you will need to be within 30-60 minutes of the CAR T center. You will also need to be monitored closely and should have your caregiver with you 24/7. You will continue to need caregiver support up to 8 weeks after the CAR T infusion. After 8 weeks, your doctor will let you know if you can restart some of your regular daily activities like driving and using heavy machinery or going back to work.
Some patients get severe side effects, while others experience mild or no side effects at all. It is important to note that side effects do not predict whether the CAR T treatment is working. If you have severe side effects, it does not necessarily mean the treatment is working. If you have mild side effects, it does not necessarily mean the treatment is not working.
Patients who start CAR T cell therapy will be told about all of the side effects they might have. It is very important that patients tell their doctors about any side effects they experience. Most side effects can be managed if they are treated early
Cytokine Release Syndrome
Your new CAR T cells will start working in your immune system right away. This can cause your immune system to go into overdrive and release molecules called cytokines. This is called Cytokine Release Syndrome (CRS). Cytokines are small proteins that control blood cells and other cells in the immune system. If your immune system releases a lot of cytokines, you may develop flu-like symptoms, such as:
- Fever
- Tiredness
- Muscle aches and pains
- Low blood pressure
- Dizziness
- Shortness of breath
- Increased heart rate
- Less urine than normal
- Body chills
- Night sweats
- Abnormal liver function
- Blood clots
Neurotoxicity
Cytokines can also affect your brain and nervous system. This side effect is known as neurological toxicity or neurotoxicity. Any damage caused by neurotoxicity can be temporary if caught early. You may not know or remember that you are having the symptoms of this side effect, but they can be very scary for your caregiver. Symptoms of neurotoxicity can vary from person to person. The severity of these symptoms may also vary depending on the type of treatment you receive. Symptoms of neurotoxicity include:
- Anxiety
- Headaches
- Sleepiness
- Confusion
- Facial droop
- Seizures
- Infection
- Tremor
- Coma
- Difficulty walking
- Change in behavior
- Change in personality
- Problems finding the right words
- Difficulty carrying a coherent conversation
- Seeing things that aren’t there (hallucinations)
Trouble keeping attention or showing confusion
Tumor Lysis Syndrome
Tumor lysis syndrome (TLS) is a condition that happens when cancer cells break down quickly, releasing chemicals into the blood. If your kidneys cannot remove these chemicals fast enough, you can develop this condition.
Cytopenia
Another common side effect is prolonged low blood cell counts (cytopenia). This is common in the first few weeks because of conditioning (lymphodepleting) chemotherapy. However, you may have this for a longer period, which would become more obvious in the first 4-8 weeks after infusion. Developing cytopenia increases your risk of infection.
B Cell Aplasia
In some cases, the CAR T drug will also attack normal, healthy B cells in the body. This can lead to B cell aplasia (low number of B cells). B cells are a type of white blood cell in our blood that helps fight off infection to support a healthy immune system. B cell aplasia makes it harder for your body to fight germs and can increase your risk of infection. To treat this side effect, you may need intravenous immunoglobulin infusions, also known as IVIG. This is only expected to occur in cancers that form in the B cells, like myeloma and certain types of lymphomas and leukemias.
Hypogammaglobulinemia
Hypogammaglobulinemia occurs when the body has low antibody levels. Antibodies are proteins in the blood that help protect the body from infection. This side effect can lead to recurrent infections, allergies, and the development of tumors (neoplasia).
CAR T Caregivers
All CAR T patients need to have a caregiver, or a trusted team of rotating caregivers, who can be with them 24/7, for at least 4-8 weeks. This person could be a family member or a friend. For some, this might be more than 1 person. If you do not have a caregiver who can stay with you 24/7 for 4 weeks, you may have multiple caregivers who each stay with you for parts of the 4 weeks. If you do not have any caregivers who can stay with you, talk to the social worker at the CAR T center about what other options you have. You may be able to find and hire a healthcare aide to stay with you. After the 4 weeks following the CAR T infusion, you may still need caregiver support, as you will be unable to drive until 8 weeks after your CAR T infusion.
The CAR T treatment team will meet with your caregiver to talk about their role and responsibilities. Most often, centers require that your caregiver is:
- 18 years old or older
- Someone who can commit to be with you 24 hours a day, 7 days a week
- Able to understand what is required of them
- Someone who is responsible and who you can count on
- In good health
- Available for 4-8 weeks
- Able to line up a backup person they can call if something happens to them
The treatment team will want to be sure that your caregiver has certain skills and abilities so they can observe any side effects that you may have and best help you during this process.
To best support you, your caregiver is going to need their own community of support. For caregivers, this could be reaching out to trusted friends or family members for support. Ask the healthcare team if they can recommend any caregiver support groups as well. Talking with a support group, a counselor, or your local CSC or Gilda’s Club location can provide advice on how to handle the cancer experience. Caregivers can also connect with other caregivers to exchange ideas and coping strategies on CSC’s Caregiver Support discussion forum through MyLifeLine.org.
Discover more resources to support caregivers.
Managing Costs
Right now, CAR T cell therapy is a very complex and expensive cancer treatment. If you do not live near a healthcare center that provides this treatment, talk with your care team about any available patient assistance programs to help cover travel costs. You can also ask if you can get assistance to cover lodging and food-related costs for your caregiver. CSC’s toll-free Cancer Support Helpline at 888-793-9355 can also help you learn more about additional financial assistance resources available to you.
Talk with your insurance to see if they will cover the drug and hospital costs. You can also ask your care team if you qualify to get this therapy as part of a clinical trial, where the drug cost is covered.
For more help on coping with cancer costs, visit our webpage about managing cancer costs.
CAR T Clinical Trials
Clinical trials are research studies to test new treatments or learn how to use existing treatments better. They find new and better ways to prevent or treat cancer. The goal of cancer clinical trials is to improve treatments and the quality of life for people with cancer. Your doctor may refer you to a CAR T clinical trial, or you may be interested in finding one on your own.
CAR T clinical trials can test different things.
- Some clinical trials are testing if an already approved CAR T therapy works in other types of cancers.
- Some may be testing a new type of CAR T therapy.
- Depending on the type of cancer you have, clinical trials may be the only way for you to receive CAR T therapy.
- There are also clinical trials to better understand how to manage CAR T side effects.
Even if you are not eligible for an approved CAR T treatment, you may be eligible for a clinical trial. Find a full list of these trials.
Frankly Speaking About Cancer Library
LibraryWould you like a print copy of these educational materials?
We can mail our Frankly Speaking About Cancer pieces to you. Shipping is free for up to 20 pounds.